Clinical & Research Highlights
Confronting Solid Tumors
Vitamin C Takes on the KRAS Gene

Dr. Manish A. Shah
About 40 to 50 percent of colon cancers have a mutation in the RAS pathway, which includes the KRAS, NRAS, and BRAF genes. A mutation in this pathway makes the disease more aggressive and less likely to respond to current therapies. With support from Stand Up 2 Cancer, Weill Cornell researchers have begun a clinical trial to validate their earlier lab findings in cell culture and in mouse in vivo studies that showed that high doses of vitamin C impaired the growth of KRAS mutant and BRAF mutant colorectal tumors. In fact, by taking advantage of the metabolic derangements caused by their specific cancer mutations, high doses of vitamin C were specifically lethal to KRAS mutant or BRAF mutant colon cancer models.
The phase 2 pilot study evaluates high dose intravenous vitamin C in two cohorts. The first cohort is of patients with resectable solid tumor malignancies likely to harbor mutations in KRAS or BRAF – for example, colon, pancreatic, and lung cancers – about to undergo surgery. Tumor samples collected during their surgery will undergo extensive genetic sequencing and molecular characterization, including development of organoid models. The organoids will be treated with vitamin C ex vivo to see if the response replicates the patient response and to determine, by manipulating the organoids, if they can overcome resistance as well.

Patients with metastatic refractory KRAS or BRAF mutant tumors make up the second cohort. They receive the same infusion of vitamin C for up to six months, with a three-month check to evaluate disease response. Information from both cohorts will be used to determine how patients with KRAS and BRAF mutations responded to treatment compared to those without those mutations. If a clinical benefit is indicated, additional trials will be designed to determine optimal efficacy and dosage, and to compare therapeutic results against other treatments, such as current chemotherapy-based standard-of-care or combination therapy.
A Microbiome Context
Weill Cornell researchers are pursuing a better understanding of the gastrointestinal microbiota, evaluating the relationships among microbiome composition, host immune response, and genomic characterization. Using whole-genome sequencing of endoscopic biopsy samples from 87 people, they compared the bacterial makeup of those with a history of H. pylori infection – including those who were treated, those with active infection, and those who developed gastric cancer – and those with no history of infection. They found that those with active H. pylori infections had less bacterial diversity, but a higher abundance of H. pylori itself. Other patients without H. pylori had more bacterial diversity. And in the cases of those with gastric cancer, most of the bacteria were found not in the tumor, but in the area immediately surrounding it. The researchers will now test 200 patients, retesting infected patients a few months after treatment with antibiotics, as well as cancer patients who have undergone immunotherapy treatments such as PD-1 inhibitors, to see how the microbiome composition changes and how it may in°uence therapy. They are also analyzing immunological data they were able to collect using the mucosal biopsies, which has shown a possible association with activation of T-cells. They hope to uncover the link between the microbiome and cancer, as well as causal immunity and tumorigenesis.
Multiple Perspectives on Breast Cancer
The p53 Gene

The p53 gene – a well-studied marker in breast cancer – is an essential pathway for tumor suppression, and mutations in the tumor suppressor gene p53 are present in up to 25 percent of primary breast carcinomas. With the knowledge that p53 mutations are the most common genetic lesions associated with human cancer, a major objective of molecular oncology researchers at Columbia is to elucidate the mechanisms by which p53 is regulated, identify the regulators for p53, and determine how to inactivate the negative regulators. The researchers have identified most of the molecules and pathways that regulate p53 activity, and current research focuses on uncovering novel strategies to target this critical pathway therapeutically, including dissecting the roles of cell metabolism in tumor suppression.
BRCA1 and BRCA2 Genes
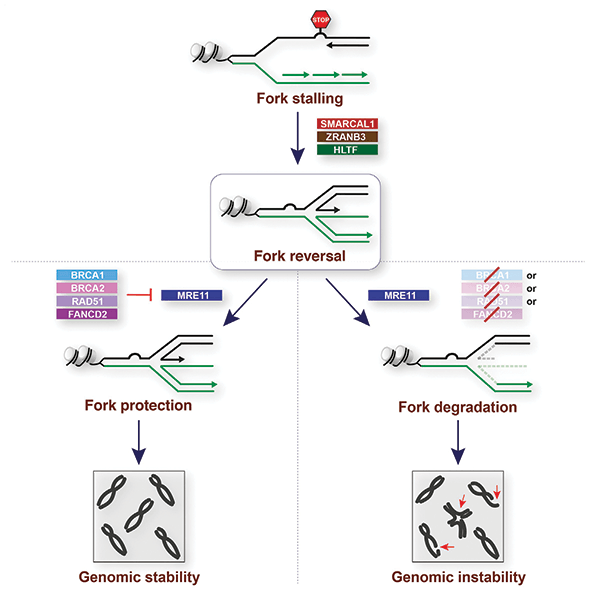
While it is well-established that mutations in the BRCA1 or BRCA2 genes can lead to breast or ovarian cancer, researchers are just beginning to decipher the mechanisms that control this process. Several studies conducted over the two decades since these genes were identified have focused on characterizing their function, including the maintenance of genome stability. Recent studies have described the role of BRCA1 and BRCA2 in promoting the stability of replication forks, structures through which the replication of the genome occurs, suggesting that a deficiency in this function can lead to chromosomal aberrations that can ultimately incite cancer development.
Studies by Columbia researchers raise the possibility that inhibition of factors that cause fork instability could reduce the occurrence of breast and ovarian cancer in BRCA1/2 mutation carriers. The Columbia research team is interested in understanding the pathways through which BRCA1 and BRCA2 protect the replication fork from degradation and avoid genomic instability. To this end, they have discovered a line of three newly characterized genes – SMARCAL1, ZRANB3, and HLTF – that cause the degradation of forks in cells with mutated BRCA1/2. Depletion of these factors restores fork integrity and reduces genome instability in BRCA1/2-deficient cells, thus suppressing potentially damaging DNA lesions and therefore possibly causing less of a propensity to develop breast and ovarian cancer. The team also has shown that inhibition of SMARCAL1 in BRCA1-deficient breast cancer cells caused resistance to chemotherapy, identifying a new chemoresistance mechanism in this form of breast cancer.
Beyond BRCA
For many years, scientists focused on the role of the BRCA1 and BRCA2 genes in breast cancer risk, but today some 30 to 40 additional genes are being tested for cancer susceptibility. As the field of cancer genetics has evolved, our researchers are expanding investigations in hereditary breast cancer genes utilizing next generation sequencing to identify germline mutations that will help clarify future cancer risk. As information on genetic mutations becomes more available, our clinicians are helping their patients and family members manage their cancer risk.

Dr. Katherine D. Crew
Mitochondrial DNA
Triple-negative breast cancer (TNBC) is aggressive and therapeutically challenging. Researchers at Columbia are exploring genetic ancestry testing through mitochondrial DNA (mtDNA), which is distinct from nuclear DNA as it is maternally inherited and allows for origin determination. This is one of the first studies of self-described African American, White, and Hispanic patients with TNBC to identify mtDNA patterns. The study, which included 92 patients with TNBC, indicated discordance between self-reported ethnicity and mtDNA analysis in 13 percent of patients. The highest discordance (26 percent) was noted in self-described Hispanic patients. The investigators concluded that the identification of mtDNA patterns with a predisposition toward TNBC may allow for risk stratification. Further studies that identify the impact of variation in mtDNA and its association with TNBC would assist in personalizing risk assessment, allowing for the potential development of ethnically tailored therapeutic interventions.
Novel Clinical Trials
As an I-SPY 2 clinical trial site, NewYork-Presbyterian/Columbia is participating in a multicenter trial for women with newly diagnosed operable breast cancer. The study is designed to test promising new treatments and identify whether these novel therapies are most effective in patients with early-stage breast cancer.

In collaboration with researchers in the Department of Systems Biology and the JP Sulzberger Columbia Genome Center, clinical researchers are conducting a pilot study to investigate genomic factors that are important for tumor growth with the ultimate goal of reducing the risk of breast cancer recurrence after surgery by targeting treatment of tumors according to their molecular dependencies. The study includes patients scheduled to undergo surgery for breast cancer after receiving chemotherapy treatment and who continue to have evidence of residual breast cancer. The investigators are using the N-of-1 clinical trial model to identify the tumor vulnerabilities that represent the Achilles’ heel of the tumor growth in that specific patient’s cancer from the analysis of its RNA rather than DNA. Initial results suggest that this type of N-of-1 study may enable a more precise therapeutic selection and identify treatments that are more universal than mutation-based personalized therapy.
Predicting Treatment Response
Patients with breast cancer who have a pathologic response to neoadjuvant chemotherapy have been shown to do better than those with a significant amount of cancer after treatment. In an observational study of 40 women with stage 2 to 3 breast cancer who received standard neoadjuvant chemotherapy, researchers used diffuse optical tomography (DOT) – a novel imaging modality that uses near-infrared light to assess hemoglobin concentrations within breast tumors – to determine its value in predicting response. The results showed that the two-week percent change in DOT-measured hemoglobin concentrations was associated with the pathologic response after five months of neoadjuvant chemotherapy. The ultimate hope is that DOT may help guide neoadjuvant therapy in the future.

Dr. Roshni Rao
A Role for Exercise
Patients undergoing neoadjuvant chemotherapy are traditionally directed to rest during treatment. A Columbia University researcher hypothesized that since breast cancer is linked to obesity, perhaps losing weight and exercising might have a positive impact on tumor biology. In a pilot study of 10 patients with stage 2-3, estrogen and progesterone receptor positive, with BMI >25, five patients were randomized to standard neoadjuvant chemotherapy plus a supervised intensive exercise regimen – “bootcamp” – and five received chemotherapy alone. While there were no initial differences between groups regarding tumor size, the mean Ki-67 for neoadjuvant chemotherapy plus exercise was 7 percent versus 29 percent with chemotherapy alone.
Addressing Treatment Toxicity

Oncologists at NewYork-Presbyterian/Columbia are designing a new clinical study focusing on ways to improve cardiovascular health in breast cancer survivors. The researchers are collaborating with experts in lipids, behavioral cardiology, and cardiovascular risk factor assessment as they seek to design interventions or studies to improve the global health of breast cancer patients. They are also working with neurologists on studies that are examining chemotherapy-related neurotoxicity.
Enhancing Surgical Results
In a refinement of nipple-sparing mastectomy, Columbia breast surgeons are employing a minimally invasive endoscopic video-assisted approach to better visualize the planes between the breast and muscle and between the breast and skin. This technique offers a less noticeable scar, excellent cosmetic outcomes, and high patient satisfaction.
Novel Treatments for Prostate Cancer
New Targeted Molecular Therapy

Dr. Scott T. Tagawa
In January 2017, Weill Cornell researchers began the first clinical trial in the United States that uses a radiolabeled small molecule to treat men with progressive prostate cancer that has spread beyond the prostate and is no longer responding to hormonal therapy. The researchers are using the radioactive particle lutetium-177 (177Lu) linked to the small molecule PSMA-617 to target prostate-specific membrane antigen (PSMA), a protein that is abundantly expressed in 85 to 90 percent of metastasized prostate cancers. The small molecule binds to PSMA and delivers precise radiation therapy intended to shrink the cancer – even in cases in which cells have yet to form a visible tumor on a bone or CT scan.
The trial primarily seeks to determine the highest dose level of the drug that can be given without significant side effects. Once the optimal dose is found, the trial will be expanded to other centers to test efficacy. PSMAtargeted therapy is thought to be one of the most promising approaches in treating metastasized prostate cancer. While this trial is the first of its kind in the United States, this same approach to treat metastatic prostate cancer has gained traction in recent years in Germany. German physicians have published anecdotes where 177Lu-PSMA-617 can reduce the volume of tumors in the body and lead to remission of the cancer. Weill Cornell researchers have been at the forefront of PSMA-targeted radionuclide (including 177Lu) therapy for more than a decade, developing the first monoclonal antibodies that could bind to PSMA in viable prostate cancer cells.

As a result, PSMA has become recognized as the best-known prostate-cancer specific cell surface molecular target. The lead antibody they developed, J591 – available nowhere else – was shown to be able to target virtually all prostate cancers in patients while also avoiding healthy tissue and normal organs, producing better responses with fewer side effects. In another trial that began in October 2017 with support from the Weill Cornell prostate cancer SPORE, the researchers are testing the effects of the more potent alpha-emitter actinium-225 (225Ac) linked to J591 in patients with prostate cancer to and the highest dose that can be administered without causing severe side effects. Weill Cornell received a $1 million Prostate Cancer Foundation Challenge Award to expand these clinical trials in several centers nationwide to test the efficacy of these highly promising new PSMA-targeted radiation-emitting treatments.
New Targeted Molecular Therapy
NewYork-Presbyterian/Weill Cornell urologists are offering high-intensity focused ultrasound (HIFU) for the treatment of localized prostate cancer. This approach – which is in its early stages of use in the U.S. – aims to preserve noncancerous prostate tissue and minimize damage to surrounding healthy tissue or organs, providing the potential for better functional outcomes. The procedure, which uses an ultrasound-guided transducer to ablate the cancerous tissue, is radiation-free and offers significantly lower risk of side effects than radiation therapy. Focally treating areas of prostate cancer can delay surgical or whole-gland irradiation, which have greater risks of erectile dysfunction and urinary incontinence.
HIFU was FDA-approved for ablation of prostate gland tissue in 2015, but not for a prostate cancer indication, citing there was not enough long-term evidence for efficacy or patient benefit. In fact, a review of studies undertaken by Weill Cornell researchers supports this concern. The investigators reported that early evidence suggests that partial gland ablation is a safe treatment option for men with localized disease. However, longer-term studies are needed to evaluate its efficacy and functional outcomes, to be able to identify optimal candidates for this therapy, and to provide data that will allow for better comparison between studies and among treatment modalities.
Cryotherapy: An Alternative Focal Therapy
NewYork-Presbyterian/Weill Cornell physicians have performed the first in-office MRI-ultrasound fusion-guided cryotherapy in New York City and the northeast. Building upon the technology that allows more accurate detection of prostate cancer during biopsy, they use cryotherapy to destroy areas of biopsy-identified prostate cancer with MRI guidance. A benefit compared to HIFU is the realtime monitoring of the area that is being thermally ablated and no limitation in terms of the prostate size.
PSMA PET/CT Scanning
NewYork-Presbyterian is among the few institutions in the world to offer PSMA PET/CT imaging, a relatively new imaging technology that greatly assists in localizing the extent of prostate cancer, as well as cancer in the lymph glands and bones. PSMA PET/CT scanning offers high sensitivity and specificity and is much more accurate than other scanning methods described to date. As a result, therapy can be targeted more appropriately.



