The 2021 Innovations in Review of clinical and scientific advances in neurology and neurosurgery highlights the life-changing and life-saving medical and surgical interventions provided at NewYork-Presbyterian Hospital. Clinicians and scientists at Columbia and Weill Cornell Medicine continue to pursue greater understanding of the complex nature of disorders of the brain and spine and define newer and more potent techniques and therapies that ease the burden of illness and hopefully effect a cure.
NewYork-Presbyterian Hospital’s neurology and neurosurgery programs – ranked #2 in the country by U.S. News & World Report – make it a destination for patients from around the world.
Breaking Barriers: Evaluating Focused Ultrasound for Drug Delivery to the Brain
The Brain: A New Normal After Menopause
A Model Way to Detect Delayed Cerebral Ischemia
Furthering Awareness in Recovery of Consciousness
Promising ALS Therapy Moves Closer to the Clinic
Spinal Cord Injury: Augmenting Function with Cervical Spine Mapping and Stimulation
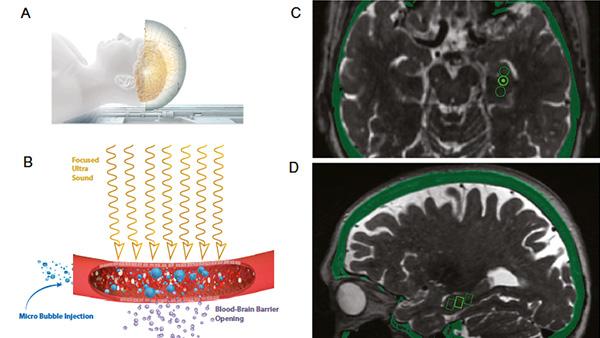
Breaking Barriers: Evaluating Focused Ultrasound for Drug Delivery to the Brain
NewYork-Presbyterian Hospital’s neurosurgeons are applying their expertise in focused ultrasound to penetrate the blood-brain barrier (BBB) and provide new pathways for drug delivery to extend and improve life for patients with glioblastoma and Alzheimer’s disease. Weill Cornell Medicine neurosurgeons injected microscopic bubbles into the bloodstream of six patients with early Alzheimer’s disease. They then directed beams of focused ultrasound to the hippocampus/entorhinal cortex, causing the microbubbles to oscillate and undergo acoustic cavitation. This induced a transient opening of tight junctions in capillaries that opened the BBB. At Columbia, a research team used focused ultrasound and microbubbles to open the BBB in a syngeneic glioblastoma mouse model to enhance the delivery of systemic etoposide, an anticancer chemotherapy, to test its effects on local tumor growth and overall survival. MRI and passive cavitation detection demonstrated the safe and successful use of focused ultrasound to open the blood-brain barrier. The combined treatment of focused ultrasound and etoposide decreased tumor growth by 45 percent and prolonged median overall survival by six days representing a 30 percent increase.
Researchers at Weill Cornell Medicine and Columbia are using focused ultrasound to facilitate drug delivery through the blood-brain barrier.
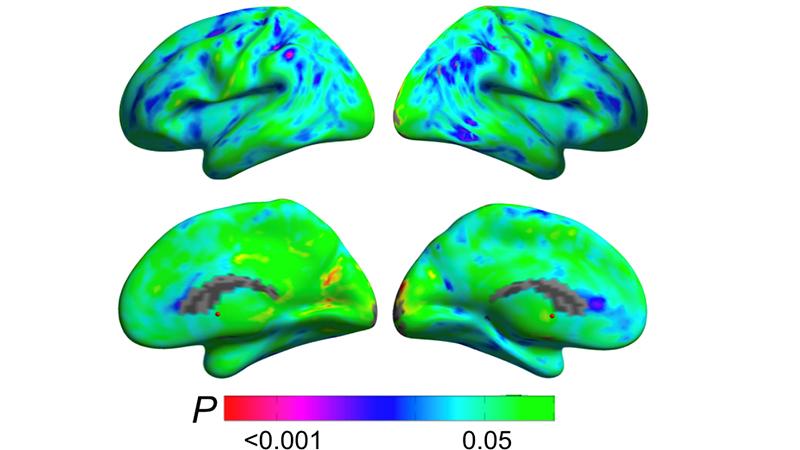
The Brain: A New Normal After Menopause
Weill Cornell Medicine neurology faculty reveal menopause is a dynamic neurological transition that significantly impacts brain structure, connectivity, and the metabolic profile during midlife endocrine aging of the female brain. The researchers used multimodality imaging to better understand the transition from pre- to post-menopause, with its steep decline in estrogen production. The study involved brain scans of women pre-, peri-, and post-menopausal. The results suggest that changes occur in many brain areas throughout the menopause transition. These include lower gray matter volume on average from the pre-menopausal to the post-menopausal group in the inferior temporal gyrus. A decline in glucose use was also noted from pre- to post-menopause over large portions of the temporal lobes as well as a decline in white matter volume in multiple regions throughout the brain. This study shows how menopause reshapes the landscape of the female brain and hints, at least, that this reshaping includes compensatory adaptations that maintain brain function despite the menopause-related drop in estrogen levels.
Brain regions showing metabolic adaptation during the transition to menopause (Courtesy of Dr. Lisa Mosconi)
A Model Way to Detect Delayed Cerebral Ischemia
Neurologists and neurosurgeons at NewYork-Presbyterian/Columbia University Irving Medical Center have developed a classification model to alert clinicians to delayed cerebral ischemia (DCI), helping to provide an opportunity to reduce neurological injury following an aneurysmal subarachnoid hemorrhage. As there are no existing monitors for DCI, the Columbia team sought to identify an alternative approach to diagnosis by applying machine learning models for DCI classification that would involve the development of hourly risk scores. The model was based on vital sign measurements and demographics routinely collected for clinical care cross-referenced with 22 time-varying physiological measures. Computation of data from patients with subarachnoid hemorrhage included mean, standard deviation, and cross-correlation of heart rate time series with each of the other vitals to explore optimal machine learning models for DCI classification. This is the first study to show that real-time hourly risk scores can classify DCI with good accuracy following an aneurysmal subarachnoid hemorrhage and that an automated, continuous monitoring tool has the potential to provide ongoing real-time risk assessment for DCI that can be implemented in any ICU.
Up to every third patient with an aneurysmal subarachnoid hemorrhage will present with delayed cerebral ischemia that can lead to devastating functional and cognitive consequences.
Furthering Awareness in Recovery of Consciousness
During the past two decades, significant advancements have been made in understanding the mechanisms underlying disorders of consciousness (DoC) caused by severe brain injuries. Neurologists and critical care medicine specialists at NewYork-Presbyterian/Columbia University Irving Medical Center and NewYork-Presbyterian/Weill Cornell Medical Center offer highly specialized expertise to promote the best outcomes in patients with DoC, an area in which there is little information on prevalence, phenotyping, and prognostication. In a review published in the March 2021 issue of Nature Reviews Neurology, the faculty at Columbia and Weill Cornell Medicine look at the mechanisms of DoC, discuss developments in detecting and predicting recovery of consciousness, and describe pharmacological and electrophysiological therapies that are offering new hope for patients. By improving understanding of the underlying mechanisms for each disorder of consciousness, patients who are in a state of cognitive motor dissociation could have a trajectory to recovery.
Columbia and Weill Cornell Medicine faculty are studying the mechanisms of recovery from disorders of consciousness following traumatic brain injury.
Promising ALS Therapy Moves Closer to the Clinic
NewYork-Presbyterian/Columbia University Irving Medical Center is leading a global phase 1-3 clinical trial of an experimental drug – jacifusen – that may alter the progression of amyotrophic lateral sclerosis (ALS). This new trial is based on promising results from a study at Columbia published in the January 24, 2022 issue of Nature Medicine that demonstrated that jacifusen lowered levels of FUS, a toxic protein, in an animal model. Columbia doctors had used the drug previously in a last-resort treatment for a young woman who had juvenile ALS, a rare and aggressive form of the disease that can affect individuals in their teens or 20s. Her disease was already advanced by the time she began the treatment and she died about a year later. However, the Columbia researchers observed that jacifusen had a profound effect, virtually eliminating the toxic proteins in her central nervous system and reducing the burden of FUS pathology dramatically. The clinical and animal data suggest that the drug has the potential to delay or prevent ALS caused by mutant FUS before symptoms appear or to slow clinical progression after disease onset.
Mutant FUS protein is toxic to motor neurons, suggesting that lowering FUS levels by silencing the gene that makes the protein might protect neurons in ALS patients with the mutation.

Spinal Cord Injury: Augmenting Function with Cervical Spine Mapping and Stimulation
A neurosurgeon at Weill Cornell Medicine is collaborating with a neuroscientist at Columbia on novel approaches to improve recovery of patients with spinal cord injury with myelopathy. Their work begins in an animal model and continues through to clinical studies to evaluate and refine an approach that uses paired brain-spine stimulation to facilitate recovery of motor function. In an experimental model, cranial stimulation paired with ipsilateral dorsal root entry zone stimulation at the level of the cord demonstrated the most significant effect on augmenting the response that is measured in muscle. In preparing for related clinical studies, cervical spine mapping was employed to guide the stimulation threshold needed to achieve a response. In a preliminary study of 15 patients, 12 had responses. All patients had undergone a posterior decompression. In the majority of the patients, the triceps gave reliably the most vigorous response, and comparing medial to lateral stimulation as measured in the triceps, lateral stimulation generated greater motor evoked potentials.
A cervical cord mapping protocol was introduced to aid in performing paired stimulation.



