Changing the Paradigm in Oncologic Imaging for Precision Medicine
Quantifying Imaging in Treatment Decision-Making
A Shift from a Qualitative to Quantitative Science
The goal in oncologic imaging is to determine how to derive the most important, biologically meaningful information from an imaging study so that a patient’s treatment can be personalized and optimized. With today’s imaging modalities, radiologists can “visualize” specific cancer disease pathways — either the interruption of those pathways or the result of the interruption of those pathways — very effectively.

Akiva Mintz, MD, PhD, Vice Chair, Research, Radiology, and Lawrence H. Schwartz, MD, Chair, Radiology, Columbia University Irving Medical Center
Supported by a robust grant portfolio in artificial intelligence (AI) and some 10 researchers, led by Binsheng Zhao, DSc, dedicated to AI endeavors, the Department of Radiology at Columbia seeks to make imaging a more data-driven quantitative science rather than a descriptive, qualitative science. The goal is to be able to more accurately interpret larger quantities of imaging data from multiple imaging modalities, such as CT, MRI, and PET, and then incorporate this information into the overall patient portfolio of other biomarkers and other personalized data points. This approach has tremendous advantages, allowing the imaging data to be more easily accessed, incorporated into algorithms, and analyzed for optimal therapeutic guidance for the patient.
“Cancer imaging is no longer just about the technology of the image acquisition; it’s also about the expertise of post-processing of the image and image analytics.”
— Lawrence H. Schwartz, MD
Oncologic imaging is performed to detect and characterize cancers, stage the disease, and assess a patient’s response to a specific therapy. Different imaging modalities and analytic techniques are used with different cancers and for these various purposes. In particular, Columbia researchers are focused on new computational and functional techniques that employ physiologic imaging and advanced image processing to assess and correlate imaging characteristics with molecular features of disease processes. While at first visual review, a mass in the lung may be benign or malignant, there are imaging features which can be measured and quantified that provide a better certainty if such a mass is cancerous. In addition, quantitative imaging may be able to accomplish much more. For example, if the same mass is determined to be cancer, there may be other features in the mass which by combining with AI techniques could reveal much more information, such as a certain mutational pattern to that tumor, or distinguish an EGFR-mutant tumor from a wild-type tumor. While this approach will not replace the role of pathology, it will help guide surgeons or interventional radiologists as to where and when to biopsy a lesion. Additional potential applications include combining an imaging study with a liquid biopsy in place of a tissue acquisition if the tumor is difficult to reach surgically or via an interventional procedure. Most important, the imaging study and the image analytics may be repeated noninvasively throughout the course of a patient’s treatment to specifically guide therapy.
Monitoring treatment response and overall survival is another key area in cancer imaging. Imaging can help clinicians and researchers determine quite early if a therapy is working for a single patient, or in a clinical trial for a group of patients. As cancer becomes more of a chronic disease, there will likely be a need for earlier imaging biomarkers to indicate the efficacy of new cancer therapies.
Columbia radiologists are now studying different imaging modalities and aspects of imaging using computational methodologies to assess tumor burden as a way of determining the optimal biomarker to investigate how a patient will ultimately do on a given therapy. This encompasses not only classes of therapy — surgery, radiation, chemotherapy — but also types of therapy, for example, combination chemotherapy or immunotherapy.
PET Imaging: Tracking the Cancer Culprit
Positron emission tomography (PET) scanning is a highly sensitive methodology of detecting molecular signatures of disease. This approach, with a goal of personalizing cancer therapies using noninvasive molecular imaging, is the subject of significant translational research endeavors by Columbia radiology faculty. These pursuits include the development of compounds that image microtubules. The Columbia team, in collaboration with colleagues at the New York State Psychiatric Institute and Wake Forest Medical Center, was the first group to identify a blood-brain barrier penetrant microtubule PET ligand. This and other innovative tracers are now being prepared for first-in-human clinical trials.
In their work on image-guided drug development, they are radiolabeling different agents to investigate their biodistribution, which is particularly significant in brain cancer because of the blood-brain barrier.
In one project, the researchers partnered with a team from Columbia Bioengineering and are employing highintensity focused ultrasound (HIFU) to open the blood-brain barrier. They have been able to demonstrate real-time distribution of various drugs with PET imaging. This is extremely important not only for diagnostic purposes, but also for demonstrating the effectiveness of the strategy to deliver the drug to its target location. They are also collaborating with Columbia neurosurgeons in imaging the distribution of radiolabeled drugs during locoregional delivery.
Novel Therapeutics
Fusion Biopsy of the Prostate and Focal Therapy of Prostate Cancer
Sven Wenske, MD, leads the Prostate Fusion Biopsy and Active Surveillance of Prostate Cancer Program, as well as the Prostate Ablation and Focal Therapy Program, in the Department of Urology at Columbia.
Fusion technology in which MRI images are merged with real-time ultrasound imaging has increased the predictive accuracy for detecting significant prostate cancer that should be treated versus prostate cancers on the indolent spectrum that suggest surveillance. Fusion biopsy allows urologists and radiologists to blend MRI and ultrasound imaging to identify areas of potentially aggressive disease and target biopsies with greater precision.
Fusion biopsy allows urologists and radiologists to blend MRI and ultrasound imaging to identify areas of potentially aggressive disease and target biopsies with greater precision.
One of the most exciting recent advances in prostate cancer diagnosis and research, the technology combines multi-parametric MRI with real-time ultrasound to provide a 3D visualization of the prostate that enables physicians to track and target suspicious areas during prostate biopsy. Because it is more accurate than other methods, MRI-ultrasound fusion has the potential to help diagnose prostate cancer with greater accuracy and avoid unnecessary invasive biopsy procedures. This technology is not only helpful in targeting lesions more accurately and identifying patients that are candidates for surveillance of their cancer, it also identifies patients that might be candidates for ablative therapies for their prostate cancer, and patients that might benefit from focal treatment of their lesion only. With advancements of the prostate MRI/ultrasound fusion technology, lesions will be able to be focally targeted and ablated under MRI-guidance.
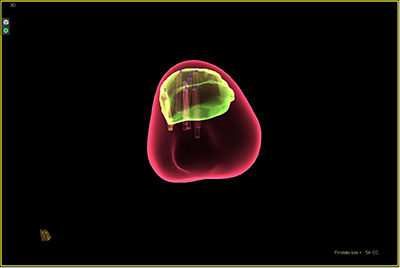
The prostate fusion biopsy system is also a research tool for urology faculty in the Irving Cancer Research Center at Columbia, who are utilizing tissue obtained through prostate fusion biopsy to further the understanding of prostate cancer biology and treatment response to different medications in vitro. Prostate fusion biopsy is pivotal to this research, facilitating the ability to identify and obtain viable tumor tissue samples that can then be tested with current and new therapies to monitor the response and growth progress of prostate cancer and allow for the development of new drugs that would circumvent resistance to treatment.



