Weill Cornell Registry to Track IBD Patient Outcomes after Transition to Adult Care
Over the last two decades, pediatricians have recognized adolescent patients with chronic disease are often inadequately prepared to manage their own medical care upon reaching adulthood. These adolescent patients have demonstrated lower rates of medication adherence and clinical visit compliance across many pediatric subspecialties and are often ill-equipped to navigate the insurance landscape and healthcare system. This lack of preparedness increases their risk of worsening disease, which can lead to poorer clinical outcomes.
Implementation of clinical transition programs and research regarding formal transition programs is scattered. Differing program structures, lack of evidence-based recommendations, varying research methodologies, and limited resources prevent the development of these programs, especially for adolescents with IBD. More prospective and outcomes research is necessary for improvement of transition care. Physicians and researchers in the Division of Pediatric Gastroenterology and Nutrition at the Komansky Center for Children’s Health are looking to provide new insights by creating a research registry to accompany their IBD Transition Program.
Dr. Kimberley Chien leads the adolescent IBD Transition Program at Komansky. With a transition team that includes the patient’s primary gastroenterologist as well as a psychologist, social worker, and other providers, IBD patients are regularly assessed on areas that include psychosocial development, health literacy, disease knowledge, the insurance system and more. Starting at age 14, the transition team works with these patients for several years to ensure they will be ready to successfully transfer to an adult gastroenterologist.
“It’s all about improving our patients’ medical outcomes and quality of life,” said Dr. Chien. “If our team can empower a patient to take part in managing their care early on, they will be able to make better decisions about managing their own disease as a young adult.”
Tracking outcomes on patients passing through this new program is a priority. Dr. Chien and a research team have established an IBD Transition Program registry. Comprehensive data will be collected in the registry, including results of quality of life and transition readiness assessments, demographic data, inflammatory markers, and records of ER visits and hospitalizations. Dr. Chien is working toward inclusion of mental health screenings in the future. The registry’s goals are not only to determine the efficacy of the program at Komansky, but also share insights with the pediatric medical community beyond NYP/Weill Cornell Medicine in order to help pediatric GIs everywhere determine the best approach for transitioning adolescent IBD patients in their practice.
“By creating the registry, we hope we will understand this adolescent population further and identify factors that might play a role in determining their transition readiness,” said Dr. Chien.
NIH-Funded Study Seeks Answers to Sex Differences in Pediatric Statural Growth Impairment
Statural growth is a dynamic, highly visible indicator of overall health in pediatric patients. It is widely known that patients diagnosed with Crohn’s disease during childhood and adolescence are often shorter than their counterparts who do not have Crohn’s. However, it remains a mystery why male Crohn’s patients more frequently suffer from statural growth impairment than female patients, despite female patients frequently having a more severe course of disease. An NIH-funded study led by one of the CADC’s physicians at the Komansky Center for Children’s Health seeks to find answers to this disparity.
Dr. Neera Gupta, physician scientist at the Komansky Center for Children’s Health at NewYork-Presbyterian/Weill Cornell Medicine, has studied growth in inflammatory bowel disease for most of her medical career, including use of bone age as a key measure in pediatric Crohn’s patients and the role of IGF-1 in statural growth in young male and young female Crohn’s patients. Her current study focusing on sex differences in statural growth impairment in pediatric Crohn’s disease (“Growth Study”) may lead researchers at Weill Cornell Medicine to better understand the underlying mechanisms of growth impairment in pediatric Crohn’s disease.
“This study has the potential to change the way we care for our patients,” said Dr. Gupta. “We rely on a partnership with our families to achieve the common goal of improving the care of our patients in the future.”
Patients will be followed for two years and will meet with the Growth Study team a total of five times after an initial screening visit if they qualify for the study. These comprehensive study visits will go beyond height and weight measurement to include patient interviews on medical history and physical development, nutritional assessments and blood tests, and up to two more bone age X-rays over the course of the two years. This wide range of data points will then be analyzed by the Growth Study team.
Dr. Gupta’s Growth Study is currently enrolling through 2016. Learn more about enrollment criteria and methodologies on the Department of Medicine’s clinical trials website.
CADC Research Grant Updates: Round Two
The Center for Advanced Digestive Care provides grant funding for pilot projects by researchers at Weill Cornell Medical College, its tri-institutional partners, and beyond. The CADC recently closed submissions for its fifth round of pilot funding.
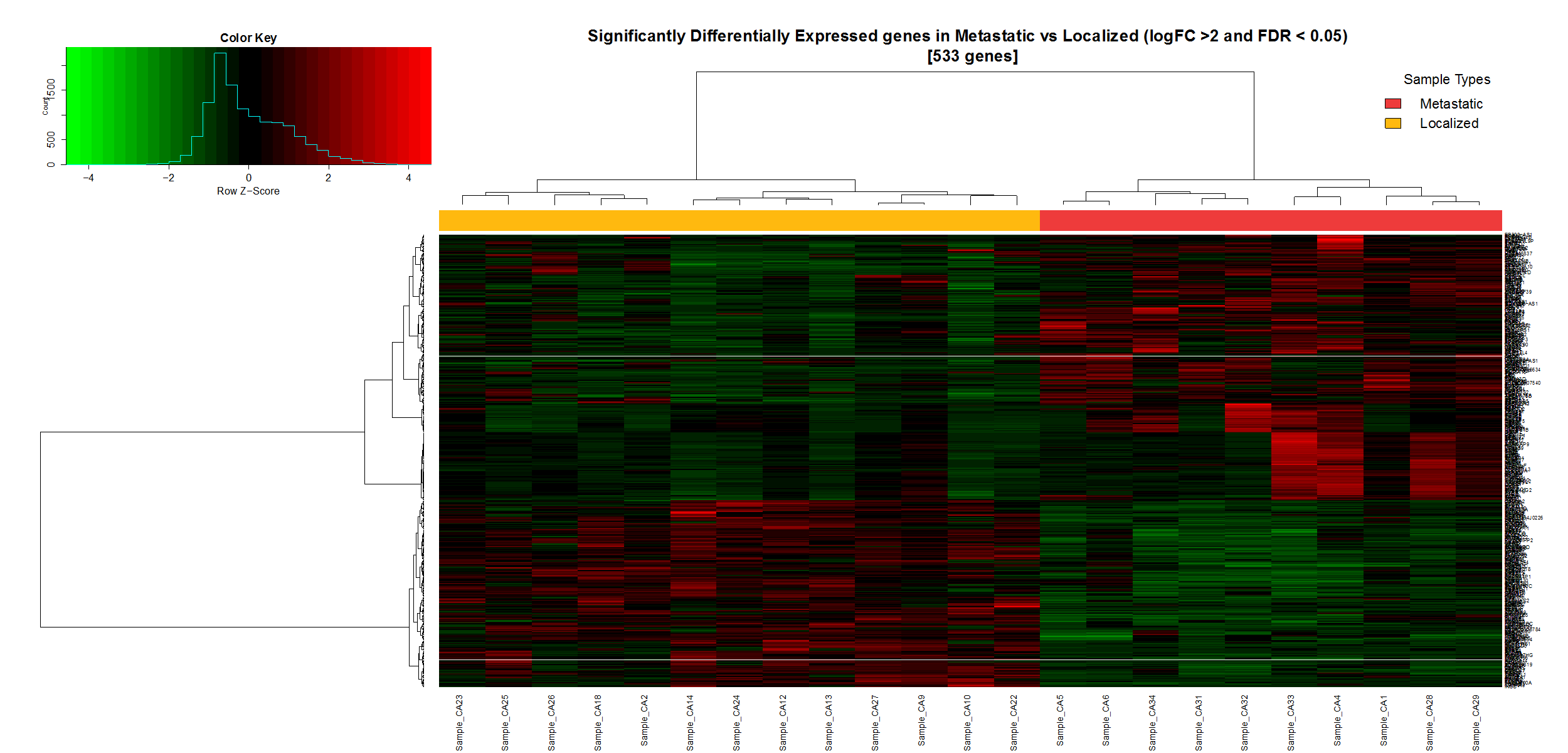
Dr. Thomas J. Fahey and a team of researchers received funding in 2014 to evaluate mRNA expressions that contribute to metastasis of pancreatic neuroendocrine tumors (PNETs). Their differential analysis compared a total of 16 localized and metastatic well-differentiated PNETs, which was an expansion of a previous study of such tumors. Several developments have come from this analysis thus far:
- Published in Annals of Surgical Oncology, the research team confirmed decreased gene expression of UCHL1 (a potential tumor suppressor) in PNET samples, as well as hypermethylation of its promoter as a mechanism.
- 533 genes were identified as being either upregulated or downregulated in metastatic PNETs. 222 of these were downregulated, and the other 311 were upregulated compared to localized primary tumors. Among these 533 significantly expressed genes, UHCL1 was confirmed to be significantly differently expressed and downregulated in metastatic tumors, which confirms the findings published in Annals of Surgical Oncology.
From this molecular profile, scientists at Weill Cornell Medicine can now identify the most differentially expressed genes between localized and metastatic tumors. The gene panel below is an example of a panel that will in the future be used to characterize the phenotype of PNETs.
RESEARCH TEAM TO STUDY MICROBIOME’S EFFECT ON GASTRIC CANCER

Helicobacter pylori (H. pylori) is the leading cause of gastric cancer manifestation, but it’s currently unclear what causes the bacterial infection to produce malignancy in the stomach. A collaboration led by Weill Cornell Medicine’s Manish Shah, MD seeks to change this by studying the gastric microbiome and its changes in the presence of H. pylori and gastric cancer.
A prevalent theory exists that H. pylori may cause chronic inflammation in the gastric mucosa, leading to a higher rate of dysplastic changes over time. With the advent of deeper research into the GI microbiome in the past few years, a second hypothesis has risen, theorizing that H. pylori may cause adverse changes to the microbiome with greater potential for malignancy. However, research on this second theory is lacking.
Along with Weill Cornell Medicine’s Doron Betel, PhD and investigators at Memorial Sloan Kettering Cancer Center and the Dana Farber Cancer Center, this research collaboration funded by the Starr Foundation hopes to build the necessary foundations for understanding the interplay between the gastric microbiome and gastric cancer. The researchers hypothesize that in the presence of a chronic H. pylori infection, host mucosal and adaptive immune systems and the microbiome are all altered, creating a perfect storm of inflammation that is ripe for malignancy.
Several examinations will take place in the course of the study:
- The gastric microbiome will be profiled in patients with and without H. pylori infections.
- The mucosal immune infiltrate will be studied in tandem with the microbiome profiles to discover consequences on host immunity.
- Genetic differences will be compared in patients with gastric cancer that developed in the setting of chronic H. pylori associated inflammation versus non-H. pylori associated gastric.
The study is projected to take two years to complete. Dr. Shah hopes that the results will form the groundwork for future microbiome work in this region of the GI tract, and even lead to improved H. pylori surveillance and treatment.
FACULTY SPOTLIGHT: PEDIATRIC GASTROENTEROLOGY AND SURGERY
In the interest of building connections and shared research opportunities between providers across NYP, the CADC Research Spotlight highlights faculty at the Center for Advanced Digestive Care, with a focus on their research and clinical interests.
This issue’s spotlight is on the two pediatric surgeons and three pediatric gastroenterologists who have joined in the past two academic years. Other recent faculty additions include Dr. Wallace Wang (gastroenterology, advanced endoscopy), Dr. Meira Ambramowitz (Jill Roberts Center for IBD), Dr. Catherine Lucero (hepatology), and Dr. Karim Halazun (hepatobiliary surgery and transplantation). These faculty members will be highlighted in future editions of Research Spotlight.

Assistant Professor of Pediatrics at Weill Cornell Medical College
Specialties
Pediatrics

Assistant Professor of Surgery at Weill Cornell Medical College
Specialties
Pediatric Surgery



