From insulin resistance to metabolic syndrome to diabetes and associated cardiac complications, cardiometabolic diseases represent a significant public health concern. At Weill Cornell Medicine, the basic science laboratory of cardiologist and physician-scientist James C. Lo, MD, PhD, is investigating metabolic conditions such as diabetes, obesity, and cardiovascular disease to better understand their fundamental molecular mechanisms with the potential of identifying novel treatment options.
Dr. James C. Lo
Diabetes is characterized by insulin resistance and insulinopenia resulting from beta cell failure and decreases in beta cell mass. In 2019, Dr. Lo and his colleagues published a paper in Nature Medicine on how healthy fat can avert diabetes. Their research, which focused on adipsin, a protein produced by fat cells, led to several avenues of investigations that yielded breakthroughs towards improved treatment for type 2 diabetes. The research also included a collaboration on human studies that involved 5,570 people from the Framingham Heart Study.
“A big problem associated with type 2 diabetes is that beta cells stop functioning properly and fade away,” notes Dr. Lo, senior author on the study. “We made the discovery that the adipokine adipsin can control blood sugar levels in a mouse model of type 2 diabetes, with a long-term positive effect on improving blood sugar and increasing insulin levels while helping to prevent beta cell death. Our findings also showed that more adipsin in the blood translated to better diabetes control.”
“Adipsin acts via the pancreatic beta cell to induce insulin secretion and protect against beta cell failure,” explains Dr. Lo. “In support of our basic research studies, we found that humans with low levels of adipsin are at higher risk of developing type 2 diabetes and beta cell failure.”
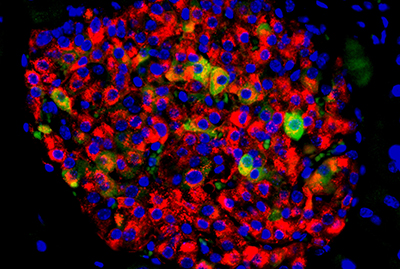
A pancreatic islet with insulin-producing beta cells (in red) and beta cells showing signs of failure (in yellow and green).
Courtesy of Lo Lab
Dr. Lo and his collaborators also studied human beta cells in their laboratories and determined that adipsin activates a molecule called C3a, which protects and supports beta cell function. They further discovered that C3a suppresses the enzyme DUSP26, which can damage beta cells and cause them to die. The researchers then directly blocked DUSP26 activity in human beta cells and found that this treatment protected the beta cells from death. Similarly, when they suppressed DUSP26 activity in mice, beta cells became healthier, meaning they could better secrete insulin.
“Some of the currently available medications that target beta cells have side effects, such as lowering blood glucose levels too much,” notes Dr. Lo. “In addition, there are no proven treatments to prevent beta cell loss. People with type 2 diabetes whose beta cells don’t work properly have to inject insulin to keep their blood glucose levels stable.”
Dr. Lo and his team are now pursuing ways to apply this novel adipose to the pancreatic islet axis to combat diabetes. Ultimately, they hope that by using adipsin or DUSP26 therapies, patients with type 2 diabetes will be prevented from developing beta cell failure and thus spared from insulin injections.