Post-Stroke Evaluation: How Safe is Transesophageal Echocardiography?
Patients hospitalized for ischemic stroke or a transient ischemic attack are already at risk for related respiratory complications – the most common causes of morbidity and mortality. Accordingly, it is essential to provide evidence whether a procedure that is beneficial for a post-stroke evaluation might also cause respiratory complications so that clinicians can make informed decisions.
Transesophageal echocardiography (TEE) is often performed in the evaluation of stroke patients to pinpoint the etiology of the stroke and potential sources of cardiac embolism. However, other than single institution case series, there is scant data provided from controlled studies supporting its safety, and some studies, although not specifically enrolling patients who had stroke or TIA, indicate the risk of complications associated with TEE may be higher than previously thought.
Dr. Hooman Kamel
Dr. Samuel Bruce
Hooman Kamel, MD, is Chief of Neurocritical Care in the Department of Neurology at NewYork-Presbyterian/Weill Cornell Medical Center and Director of the Clinical and Translational Neuroscience Unit at Weill Cornell Medicine’s Brain and Mind Research Institute. Samuel S. Bruce, MD, is a neurology resident and soon-to-be stroke fellow at NewYork-Presbyterian/Weill Cornell. Dr. Bruce and Dr. Kamel recently undertook an IDEAL phase 4 study with colleagues in cardiology and pulmonary and critical care medicine at Weill Cornell Medicine to address the gap in safety data related to TEE, with a particular focus on severe but rare complications such as aspiration leading to respiratory failure.
While transthoracic echocardiography has the advantage of assessing patients non-invasively, TEE is often performed because it can provide better visualization of certain cardioembolic sources. In their retrospective study, the Weill Cornell Medicine research team, led by Dr. Bruce, compiled data on a random sample of Medicare beneficiaries over a nine-year period from inpatient and outpatient insurance claims collected by the Centers for Medicare and Medicaid Services. The study population included patients 65 years or older hospitalized between 2009 and 2018 for acute ischemic stroke and TIA with a primary discharge diagnosis of stroke documented upon admission. The principal outcome measure was acute respiratory failure defined as endotracheal intubation and/or mechanical ventilation, starting on the first day after admission through 28 days afterward.
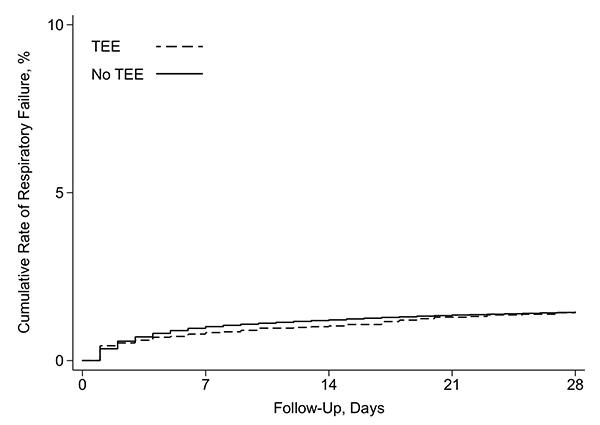
In a cohort of patients with ischemic stroke or transient ischemic attack, there was no difference in the cumulative risk of respiratory failure between those who underwent TEE (dashed line) and those who did not undergo TEE (solid line).
Characteristics of the study population and associated findings, published in the February 7, 2022, issue of BMJ Surgery, Interventions, and Health Technologies, included:
- Analysis of 99,081 Medicare beneficiaries
- 73,733 patients (74.4 percent) had an ischemic stroke, and 25,348 patients (25.6 percent) had a TIA
- TEE was performed in 4,677 patients (4.7 percent): 4,137 with an ischemic stroke and 540 with a TIA
- Patients undergoing TEE were younger, more often male, less often had atrial fibrillation and congestive heart failure, more often had valvular heart disease and infective endocarditis, and had higher rates of treatment with thrombolysis, thrombectomy, and cardioversion
- 1,403 patients (1.4 percent) underwent intubation and/or mechanical ventilation during the study period
- Patients who had respiratory failure were younger but had more vascular risk factors and comorbidities than those who did not require intubation or mechanical ventilation
Importantly, the researchers found – across multiple sensitivity analyses – that the cumulative risk of respiratory failure at 28 days in patients who underwent TEE was similar to those who did not undergo TEE. These results can help clinicians in their decision making on whether to perform TEE for a post-stroke etiologic evaluation.
Read More
Transesophageal echocardiography and risk of respiratory failure in patients who had ischemic stroke or transient ischemic attack: an IDEAL phase 4 study. Bruce SS, Navi BB, Zhang C, Kim J, Devereux RB, Schenck EJ, Sedrakyan A, Díaz I, Kamel H. BMJ Surgery, Interventions, and Health Technologies. 2022 Feb 7;4(1):e000116.
Related Publications