The 2021 Innovations in Review of clinical and scientific advances in cardiology and cardiovascular surgery highlights NewYork-Presbyterian Hospital’s novel clinical and scientific endeavors pursued to maximize the outcomes for patients with all levels of disease complexity. The accomplishments of the Columbia and Weill Cornell Medicine heart programs are rooted in the close collaborations among faculty, who develop clinical strategies – medical, interventional, or surgical – that are best suited for the patient.
NewYork-Presbyterian Hospital’s adult cardiology and heart surgery programs – ranked #4 in the country by U.S. News & World Report – make it a destination for patients from around the world.
Wave of the Future: Remote Cardiac Monitoring for Heart Failure
Reducing Risk of Atrial Fibrillation After Cardiac Surgery
Hypertrophic Cardiomyopathy: The Role for Septal Myectomy

Wave of the Future: Remote Cardiac Monitoring for Heart Failure
NewYork-Presbyterian’s Centralized Heart Failure Management Program in partnership with Columbia and Weill Cornell Medicine is applying the latest evidence-based technology in remote cardiac monitoring to manage patients with symptomatic heart failure from their home. This approach is part of a five-phased vision of the heart failure team using patient specific data and artificial intelligence to identify and monitor patients with heart failure, and in the future prevent the condition from developing. A dedicated heart failure team partners with local doctors and monitors patients using remote monitoring technology to measure pulmonary pressure, blood pressure, weight, and other physiologic changes that are used to adjust a patient’s medical regimen. The promising approach of remote cardiac monitoring has led to the emergence of a number of devices incorporating either invasive or noninvasive technologies. A comprehensive review by Columbia and Weill Cornell Medicine faculty published in the December 29, 2021 issue of JAMA Cardiology details the current advances and the major clinical trials that are underway evaluating these technologies.
Implementation of remote monitoring technologies marks a transition in the heart failure field.
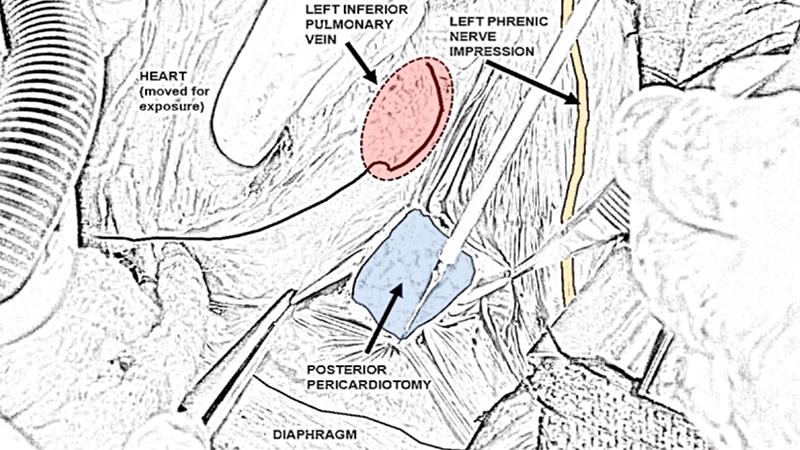
Reducing Risk of Atrial Fibrillation After Cardiac Surgery
Weill Cornell Medicine researchers have established that a procedure during cardiac surgery was associated with a 56 percent reduction in the incidence of atrial fibrillation with no added risks or side effects. Their findings in a recent study suggest that posterior left pericardiotomy has significant potential for preventing prolonged hospital stays and the need for additional interventions and drugs to reduce the risk of strokes and heart failure associated with atrial fibrillation. The study evaluated whether a posterior left pericardiotomy, a surgical maneuver that drains the pericardial space into the left pleural cavity, could help prevent atrial fibrillation in patients who underwent cardiac surgeries. Results of the randomized controlled trial were published in the December 4, 2021, issue of The Lancet. Study investigators also presented their findings in a live virtual late-breaking science session at the American Heart Association’s Scientific Sessions 2021.
The posterior pericardiotomy is a 4- to 5-cm long incision in the pericardium located posterior and parallel to the phrenic nerve between the left inferior pulmonary vein and the diaphragm. The opening allows drainage of pericardial fluid, blood, and clots into the left pleural space. (Courtesy of The Multimedia Manual for Cardio-Thoracic Surgery)

Hypertrophic Cardiomyopathy: The Role for Septal Myectomy
What are the effects of septal reduction therapy on acute cardiovascular manifestations and all-cause mortality compared to non-invasive management in patients with hypertrophic cardiomyopathy? This is the question that cardiac surgeons in the Hypertrophic Cardiomyopathy Center at Columbia sought to answer in a study – the first of its kind – published in the September 30, 2021, issue of the International Heart Journal. Some 80 percent of patients with hypertrophic cardiomyopathy (HCM) will experience mechanical obstruction to the left ventricular outflow (LVOT) resulting in cardiovascular complications. When symptoms do not respond to maximal medical therapy, septal reduction therapy (SRT) is performed to reduce LVOT obstruction. However, it is largely unknown whether this procedure prevents HCM-related cardiovascular events or death. Columbia faculty examined this issue in 752 patients with HCM who underwent SRT between 2007 and 2014 and 5,855 controls with HCM who had never had SRT. Their findings revealed that patients undergoing SRT had a reduced risk of acute cardiovascular events and mortality in the first year following surgery.
Axial MRI angiogram of cardiac hypertrophy of the interventricular septum
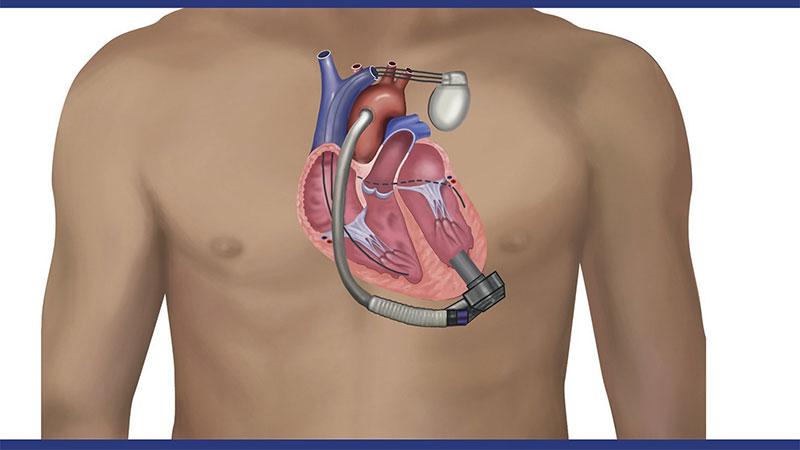
LVAD: A Change of Pace for CRT
Heart failure specialists at NewYork-Presbyterian Hospital have conducted the first prospective randomized study of cardiac resynchronization therapy (CRT) in patients with an LVAD. Their findings demonstrated that right ventricular pacing as compared to biventricular pacing significantly improves functional status and quality of life. A large number of patients with heart failure have CRT devices implanted at the time of LVAD implantation and both independently improve patient outcomes. As evidence related to the benefits of combining these therapies remains scant, Columbia and Weill Cornell Medicine cardiologists investigated the pathophysiologic mechanisms in place when these two heart failure technologies are used together. Their findings, which were published in the August 2021 issue of JACC: Clinical Electrophysiology, also showed that right ventricular pacing improved left ventricular volumes and reduced tachyarrhythmias, while keeping pacemaker lead impedance stable. In light of their findings, the authors recommend turning off LV lead pacing in patients on LVAD support with CRT devices.
Many patients with heart failure have cardiac resynchronization therapy devices implanted at the time of LVAD implantation.
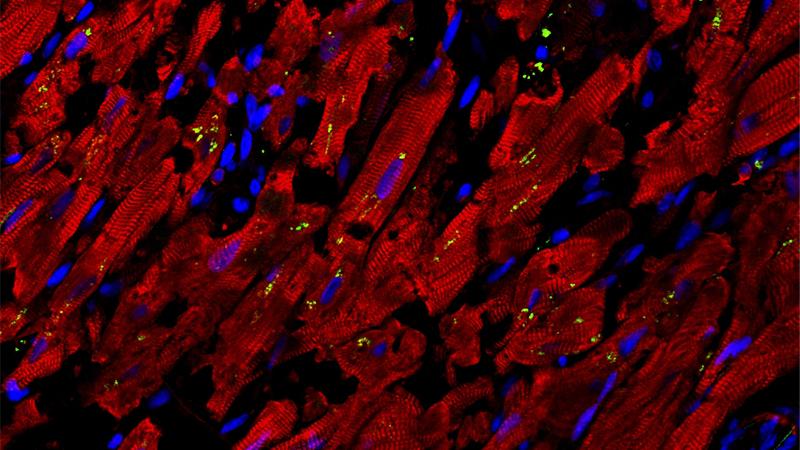
When COVID-19 Attacks the Heart
Investigations by faculty at Weill Cornell Medicine and Columbia have uncovered the source of cardiac injury in patients hospitalized with COVID-19 and identified FDA drug candidates as potential therapies to prevent or ameliorate this type of cardiac complication. Seeking to understand the cause behind the cardiac damage, which is commonly seen in some 20 to 30 percent of hospitalized patients, the researchers conducted a study of heart samples of patients with COVID-19. Their findings, published in the June 25, 2021, issue of Circulation Research, demonstrated that abnormal inflammatory infiltration of macrophages in heart tissues lead to a secretion of cell-damaging proteins. The research team then established a model system to better mimic human physiology and facilitated automated methods to quickly screen for drugs that could alleviate this cellular injury. Performing a high content screen of nearly 1,300 drugs already approved by the FDA, they found two compounds – ranolazine and tofacitinib – that robustly reduce both the levels of reactive oxygen molecules and the deaths of cardiac muscle cells.
Confocal image of heart autopsy sample of a COVID-19 patient showing the damaged structure of cardiomyocytes (red) and increased expression of chemokine, CCL2 (green), which recruits macrophages that further damage heart tissue. (Courtesy of Dr. Liuliu Yang)



