An interventional cardiology team at NewYork-Presbyterian and Columbia were among the first in the country to use a novel transcatheter valve system to treat symptomatic, severe aortic regurgitation (AR), opening the door to more optimal minimally invasive solutions for patients who cannot undergo surgery.
The Trilogy™ Transcatheter Heart Valve System was recently approved by the Food and Drug Administration as the first transcatheter solution in the U.S. with a dedicated indication for severe, symptomatic AR patients at high or greater risk for surgical aortic valve replacement (SAVR). The disease has historically been difficult to treat minimally invasively because unlike with aortic stenosis (AS), there is no calcification that traditional transcatheter aortic valve replacement (TAVR) devices can latch onto. As a result, when TAVR was previously used for AR, the valves tended to migrate — and even when they stayed in place, there was a higher risk of paravalvular leaks.
We’re very excited to finally have a new, less invasive therapy for high-risk patients with aortic regurgitation. This device allows these patients to get treated without open-heart surgery.
— Dr. Torsten P. Vahl
“Overall, the outcomes were much worse when TAVR was used off-label in aortic regurgitation patients than what we were used to from aortic stenosis patients,” says Torsten P. Vahl, M.D., an interventional cardiologist at NewYork-Presbyterian and Columbia and national principal investigator of the ALIGN-AR trial, which helped the new technology gain FDA approval. “We're very excited to finally have a new, less invasive therapy for high-risk patients with aortic regurgitation. This device allows these patients to get treated without open-heart surgery.”
How the New TAVR System Works
The system uses metallic anchors, known as locators, to clasp onto the three cusps of the aortic valve, trapping the native leaflet between the locator and the body of the device. This configuration creates an outer seal that prevents paravalvular leaks. “It clips on like three paperclips. As a result, it can't migrate into the ventricle because the locator provides a stopping point when it hits the bottom of the leaflet. And the risk of it moving into the aorta is also lower,” explains Dr. Vahl. “Once this device is in place, it can serve as an anchoring platform for additional TAVR valves to be placed inside it in the future.”
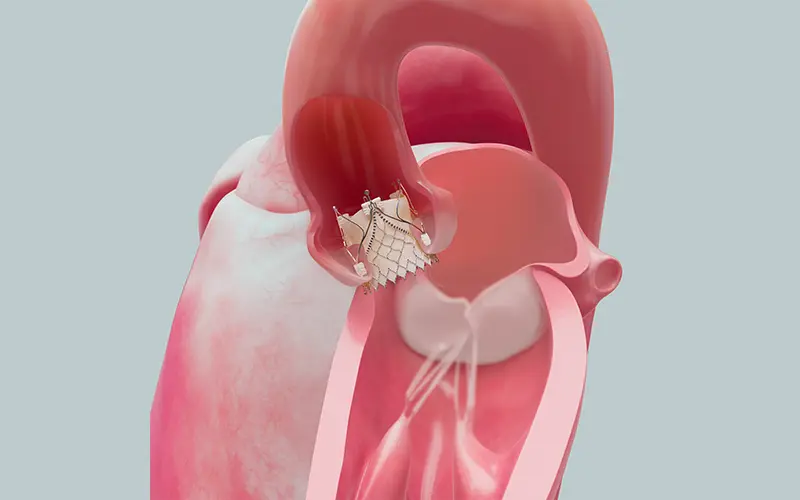
The Trilogy system uses metallic anchors to clasp onto the three cusps of the aortic valve, creating a seal to prevent paravalvular leaks. Image courtesy of JenaValve Technology.
The technology can also be used for other high-risk groups, such as patients with left ventricular assist devices (LVADs). “These patients sometimes develop aortic regurgitation and are considered high-risk for open heart surgery due to their existing LVAD,” he adds.
The ALIGN-AR Trial
The safety and efficacy of the system were established through the pivotal ALIGN-AR study, led by Dr. Vahl with study chairman Martin B. Leon, M.D., chief innovation officer and the director of the Cardiovascular Data Science Center for the Division of Cardiology at NewYork-Presbyterian and Columbia.
The first phase of the ALIGN-AR trial enrolled 180 AR patients across 20 U.S. cities who were at high risk for mortality or complications if they were to undergo SAVR. The second phase analyzed data from 700 patients (180 from the pivotal trial and 520 continued-access patients) across 30 sites. Co-primary endpoints were a composite of major adverse events 30 days post-procedure and one-year all-cause mortality.
The results, published in The Lancet, demonstrated significant improvement after transcatheter aortic valve intervention with the novel system:
- The 30-day composite endpoint was 24%, meeting the performance goal.
- All-cause mortality was 7.7% at one year, significantly lower than the target endpoint.
- Substantial reverse cardiac remodeling was observed, such as a decrease in the mean left ventricular volumes and mean left ventricular mass.
- Functional status improved in most patients, with 81% demonstrating an improvement of at least one New York Heart Association class category at one year.
Ongoing evaluations will continue to assess the long-term performance of the device and overall survival and mortality among this patient population.
Meanwhile, AR patients with a lower surgical risk should still consider SAVR, but there are new clinical trials suited to this population that are currently being conducted at NewYork-Presbyterian and Columbia. “Patients could possibly enroll in the ARTIST trial, where low- and intermediate-risk patients with significant aortic regurgitation are randomized to either open-heart surgery or TAVR with the Trilogy device,” explains Dr. Vahl.
The Next Generation of Care
Just as TAVR became a transformative, minimally invasive approach for treating AS, Dr. Vahl believes the novel technology carries the same potential for AR. “Initially, no patients with aortic regurgitation were referred to our valve clinic,” he says. “Cardiologists and primary care doctors did their best to manage heart failure symptoms medically because there were no alternative therapies available for high-risk patients. Now, we have an additional tool. This is very exciting news for our patients.”
A version of this article originally appeared on the Columbia University Irving Medical Center website.




