Glioblastoma multiforme (GBM) is the most aggressive and deadliest form of primary brain cancer, accounting for roughly half of all gliomas. Despite extensive research over the past three decades into therapies for the disease, overall life expectancy for GBM patients remains only about 15 months.
A significant challenge in treating GBM lies in the limitations of preclinical models, which fail to accurately mimic the tumor cells’ ability to migrate from the center of the tumor and infiltrate neural networks. “When I was a fellow in 1985, the median survival of a patient with glioblastoma was about 13 months. We’ve made all of two months’ progress in 40 years,” says Howard Fine, M.D., chief of the Division of Neuro-Oncology in the Department of Neurology at NewYork-Presbyterian and Weill Cornell Medicine and founding director of the Brain Tumor Center at Weill Cornell Medicine. “It was obvious that the model systems used in the field didn’t represent human disease at all, and maybe that was one of the reasons why we’ve made so little progress.”
Dr. Fine has dedicated his career to developing better therapies for GBM, and over the past 15 years has focused on creating GBM models that genetically and clinically behave like human gliomas in a patient-specific way — a journey that ultimately led to growing human cerebral organoids, also known as mini brains, in a lab. “We needed to build a model system that represented the diffuse infiltration and integration of glioma in the normal brain,” he says. “And that’s what we’ve done.”
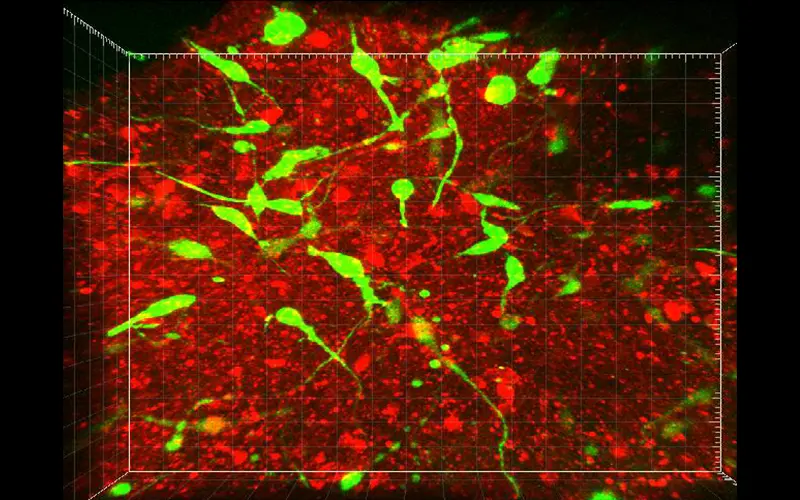
Confocal microscopy showing a brain organoid (red) and glioma cells (green) five days post co-culture.
Developing the Glioma Cerebral Organoid (GLICO) Model
The early stages of Dr. Fine’s organoid modeling research began while he was at the National Institutes of Health, when in 2006 he and his team were among the first in the world to isolate glioma stem cells and maintain them in a functional state outside the human body. “We demonstrated that these are the true progenitor cells of gliomas,” he says. “However, when you place them in a mouse brain, they don’t recapitulate the environmental effect you need that’s so important for understanding the disease and developing effective therapies.”
In 2013, researchers in Austria published a paper demonstrating the use of cerebral organoids in modeling microcephaly, “and the light bulb went off. I realized we could use this for our ultimate human glioblastoma model,” Dr. Fine says. “We ran with it, and we’ve been developing the technology ever since.” In 2017, after joining NewYork-Presbyterian and Weill Cornell Medicine, Dr. Fine received funding through an NIH Director’s Pioneer Award to further his research on using advanced stem cell techniques to grow glioma cerebral organoid (GLICO) models, a process that involves several stages:
- Human embryonic stem cells or induced pluripotent stem cells derived from patients are cultured in conditions that promote neuroectoderm formation, taking about six to eight weeks to form a mature cerebral organoid.
- The patient’s glioma stem cells (GSCs) are co-cultured in bioreactors with the fully formed organoids.
- Within 24 to 48 hours after co-culture, the GSCs begin to invade and destroy healthy neural tissue, mimicking the behavior of tumors in patients.
When cerebral organoids mature, they develop all the major cell types found in a developing human embryo, such as neurons, myelinated axons forming synapses, retinal epithelium, and the choroid plexus. Some of the cerebral organoids from Dr. Fine’s laboratory have been maintained for several years, evolving from early embryonic brains to more developed adolescent or elderly brains. A one-month-old cerebral organoid has the neurodevelopmental maturity of an 8-to-12-week-old fetus, while 8-to 12-month-old organoids resemble the brain of a 3-to-6-month-old baby. “These cerebral organoids respond to standard neurotransmitters like glutamate and serotonin just like a normal brain, aging in a way that parallels human development,” says Dr. Fine.
Another defining feature of the GLICO model is that it develops tumor microtubes — long, thin protrusions that penetrate deeply into the brain — that mimic how GBM cells naturally proliferate. “All these infiltrating tumor cells, which we always thought acted like lone rangers, are connected with each other through these tentacles, and allow communication between tumor cells and normal cells across great distances within the brain. It’s almost like an internet of tumor cells interdigitating,” explains Dr. Fine. “You could imagine why this would be so difficult, if not impossible, to treat with radiation, surgery, or chemotherapy. It’s become clear that the way we’re going to defeat this tumor is by breaking down these connections and making these cells stand alone, so to speak.”
Glioma cells communicating with one another using synaptic connectivity.
Using GLICO Models to Advance Glioblastoma Personalized Therapy
Dr. Fine and his team have published multiple papers that demonstrate how the GLICO model has effectively replicated patient-specific GBM features, as every GBM is genetically unique — something that traditional cultures have failed to do — and that offer insight into the factors that could impact drug efficacy.
We can test 1,500 FDA-approved drugs per GLICO and find the drug or drug combinations that look to be the most effective against that patient’s particular tumor, in the setting of their mini brain.
— Dr. Howard Fine
He has begun the work needed to help establish regulatory guidelines that could pave the way for a future clinical trial that uses GLICO models to provide individualized drug screening for glioblastoma patients. With funding from the NIH and the Starr Foundation, Dr. Fine and team worked with Olivier Elemento, Ph.D., director of the Englander Institute for Precision Medicine at Weill Cornell Medicine, to develop a robotic, automatic, high-throughput drug-screening system capable of screening hundreds of GLICOs against thousands of drugs to assess efficacy and neurotoxicity.
“We can test 1,500 FDA-approved drugs per GLICO and find the drug or drug combinations that look to be the most effective against that patient’s particular tumor, in the setting of their mini brain, because every glioblastoma is dramatically different,” says Dr. Fine. “This is the next generation of precision medicine.”
Dr. Fine says that within the next several years, he also expects artificial intelligence to play a pivotal role in advancing treatment by developing predictive algorithms for therapeutic response, based on data from these large screening initiatives using GLICO against several hundred unique glioma stem cell lines from well over a hundred GBM patients.
“The hope is that this model would allow us in the future to sequence a patient’s tumor, input that data into the algorithm, and get recommendations for which drugs may work best,” says Dr. Fine. “By doing this computationally, we can save the effort, resources, and time that go into generating a GLICO and doing the drug screening. It’s a very realistic goal, and one that we’re aiming for.”




