August 2, 2016
The world of clinical gastroenterology has found biologics such as infliximab to be helpful tools in treating the unpredictable relapsing and remitting pattern of inflammatory bowel disease. Unlike other classes of IBD drugs frequently administered orally or by suspension, biologics require infusions in a hospital setting or at specialized infusion centers. In a study published in the May 2016 issue of Practical Gastroenterology, three gastroenterologists from Weill Cornell Medicine and the Phyllis and David Komansky Center for Children’s Health demonstrate that a similar quality of life can be achieved if patients perform infusions at home.
Doctors Elaine Barfield, Robbyn Sockolow and Aliza Solomon used a sample of 38 IBD patients for the study, consisting of 18 subjects and 20 controls. 33 had Crohn’s disease, 3 had ulcerative colitis, and 2 had indeterminate colitis. Patient quality of life was assessed at a follow-up visit by completing the IMPACT III assessment, which evaluates quality of life across six areas: bowel symptoms, systemic symptoms, emotional functioning, social functioning, body image, and tests/treatments. The study was limited to English-speaking patients who were adherent with their infliximab regimen prior to the study, and those with an ostomy were also excluded, as the IMPACT III survey is not considered valid for measuring quality of life in these patients. Patients selected for each arm of the study completed the survey.
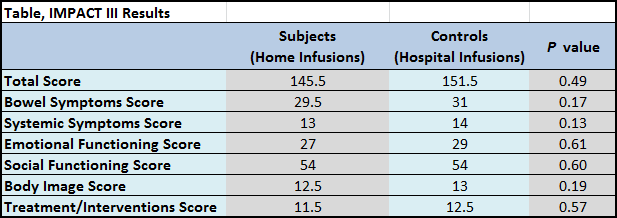
Results of the IMPACT III assessment at the end of the study showed only slight differences in quality of life between patients receiving home versus hospital infusions. With possible total scores for IMPACT III ranging from 35 to 175, the median score for the subject (home infusion) group was 145.5, while the control (hospital infusion) group’s median score was 151.5 (p=0.49). Individual measurement areas were similar, with the largest swing being emotional functioning, with 27 for those receiving home infusions and 29 for those in the control group (p=0.61). Some limitations are acknowledged in this study, including small sample size and possible recall bias when filling out the final IMPACT III surveys.
While the negligible difference between quality of life scores suggests that neither delivery method is inferior, the Weill Cornell team was surprised.
“We went into the study with a sense that quality of life in patients receiving home infusions would be greater than that in patients receiving hospital infusions, since anecdotal communication with families suggest a preference for home infusions including less anxiety and fewer work and school absences,” said Dr. Elaine Barfield.
Dr. Barfield and the other authors suggest in their paper that the location of infusions may not reduce feelings of isolation, unpredictability and loneliness associated with having a chronic disease like IBD. Furthermore, younger patients may not appreciate the benefit of traveling to the hospital and avoiding missing days of school or work comes along with home infusions. Even so, the authors see value in discussing and offering home infliximab infusions should families desire to pursue these. They also suggest that inclusion of a quality of life survey in pediatric patients may help clinicians identify patient needs unmeasured by the typical pediatric Crohn’s disease and ulcerative colitis disease activity indexes.


